您想继续阅读英文文章还
是切换到中文?
是切换到中文?

THINK ALUMINIUM THINK AL CIRCLE

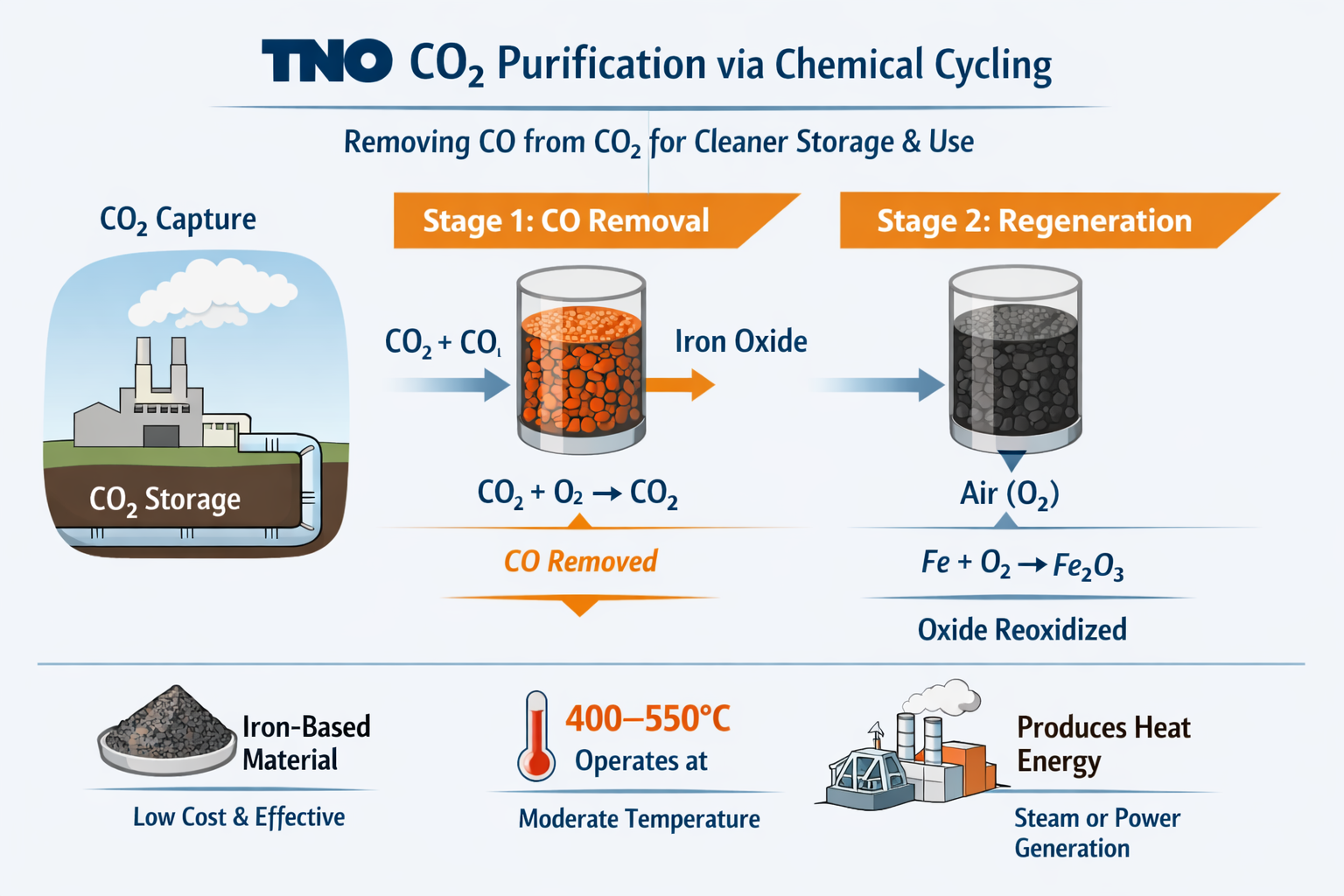
The Netherlands Organisation for Applied Scientific Research (TNO) developed a method to purify carbon dioxide (CO₂) for underground storage or industrial use. The method removes carbon monoxide (CO) from CO₂ using a process called chemical cycling.
{alcircleadd}In carbon capture and storage, captured CO₂ often contains impurities. These impurities can damage pipelines, storage sites, or catalysts used in later processes. Carbon monoxide is particularly restricted, and its concentration must generally remain below 100 to 300 ppm. Existing purification methods often require high energy, complex equipment, or may introduce additional contaminants.
For the global aluminium value-chain 2026 outlook, book our exclusive report “Global ALuminium Industry Outlook 2026"
The proposed method uses metal oxide particles as oxygen carriers in a two-stage process. In the first stage, a gas mixture of CO₂ and CO passes through the metal oxide. The oxide releases oxygen, which reacts with CO and converts it into CO₂. During this reaction, the metal oxide will be reduced partially.
In the second stage, the reduced oxide reacts with air and becomes oxidised again, restoring it for reuse in another cycle. This process removes CO without adding other substances to the gas stream.
The researchers used thermodynamic analysis to select suitable materials. They evaluated oxides of nickel, copper, manganese, and iron under temperatures below 700°C and low CO concentrations. Copper and iron oxides excelled, while nickel and manganese were less stable. Both reaction stages released heat, indicating that the process could also produce usable energy for steam or electricity generation.
Also read: Rio Tinto advances gallium extraction project in Quebec with federal backing
Iron-based materials were studied in more detail because it is widely available, non-toxic, and cost-effective. Several iron oxide materials supported by aluminium oxide were tested. The most effective sample, produced by an infusion method, operated for about 200 cycles at 400 to 550°C while converting CO to CO₂. The system also functioned effectively at CO concentrations of 2,000 to 4,000 ppm, which are typical of industrial gas streams.
Experiments showed that raising the temperature to around 450°C increased the time required for complete CO conversion, and further increases provided only limited improvement. As a result, the optimal operating temperature depends on the process design and how the released heat is used.
Don't miss out- Buyers are looking for your products on our B2B platform
Although iron-based materials performed well, other materials may offer faster reactions or greater durability. Future work will test additional materials and evaluate the technical and economic feasibility of the process compared with methods such as cryogenic separation (energy-intensive) or catalytic oxidation (chemical process using a catalyst for oxidation).
Note: The image used in this article is generated with an AI tool and does not depict any real-time moment
Responses








